 Ii) a, b, c, d Calculate the frequency of each wavelength of electromagnetic radiation:Ĭ.) 5.8 * 10^18 Hz Calculate the energy of a photon of electromagnetic radiation of each of the wavelengths:Ĭ.) 3.8 * 10^-15 J A laser pulse with a wavelength 532 nm contains 3.85 mJ of energy. How long does it take light to travel from the sun to the Earth? 499 seconds List these types of electromagnetic radiation in order of (i) increasing wavelength (ii) increasing energy per photon The energy of one photon of this light is _ J.ġ.14 × 10-31 3.46 × 10-19 The distance from the sun to Earth is 1.496*10^8 km. As the energy decreases the frequency decreases Electromagnetic radiation with a wavelength of 575 nm appears as yellow light to the human eye. The wavelength and frequency are independent of each other. Radio waves have a smaller wavelength than visible light.Īs the energy decreases the frequency decreases. The amplitude of a wave depends on the frequency. 4, -3, -2, -1, 0, +1, +2, +3, or +4 0 or 1 Determine the lowest frequency of light capable to remove an electron from a sample of Ti metal, if the binding energy of titanium is 3.14 × 103 kJ/mol.Ĥ.74 x 1015 Hz 7.87 x 1015 Hz Which statement about electromagnetic radiation is correct?Īs the wavelength decreases the energy decreases. N = 1, l = 0, ml = 0 n = 1, l = 0, ml = 0 What are the possible values of l if n = 2? N = 6 to n = 5 n = 1 to n = 2 Identify the correct values for a 1s sublevel. Calculate the ionization energy (in kJ) for a mole of hydrogen atoms, making the assumption that ionization is the transition from n = 1 to n = ∞.Ģ.76 × 103 kJ 1.31 × 103 kJ What is the energy of light associated with a transition from n=3 to n=8 in a hydrogen atom? Does this represent absorption or emission of a photon?Ģ.08 × 10-19 J, emission 2.08 × 10-19 J, absorption Choose the transition (in a hydrogen atom) below that represents the absorption of the shortest wavelength photon. Schrödinger quantum number angular momentum quantum number It is possible to determine the ionization energy for hydrogen using the Bohr equation. Ga Xe How many orbitals are present when l=3?ĩ 7 Which of the following quantum numbers describes the shape of an orbital? Which of the following elements is NOT a metal? Electrons in the 2s orbital can penetrate the 1s orbital and be closer to the nucleus. There are more nodes found in the 2s orbital. The shape of the orbital ultimately determines the energy of the electrons.Įlectrons in the 2s orbital can penetrate the 1s orbital and be closer to the nucleus. The larger number of electrons found in the 2p orbital leads to greater repulsion. 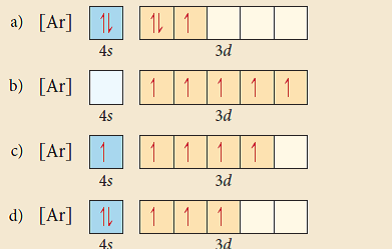  
Core electrons effectively shield outer electrons from nuclear charge Why does an electron found in a 2s orbital have a lower energy than an electron found in a 2p orbital in multielectron systems?Įlectrons in the 2s orbital are shielded by electrons in the 2p. Titanium chromium Give the ground state electron configuration for Cd.Ĥd10 5s24d10 Choose the statement that is TRUE.Ī.) Core electrons effectively shield outer electrons from nuclear charge.ī.)Valence electrons are most difficult of all electrons to remove.Ĭ.)Core electrons are the easiest of all electrons to remove.ĭ.)Outer electrons efficiently shield one another from nuclear charge.Į.)All of the above are true. The element that corresponds to the electron configuration 1s22s22p63s23p64s13d5 is _ 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
